Maritime Climate Concerns
While the oyster is center stage in global cuisine and aquaculture, there is a dark side to every success story. Our oceans, estuaries, and rivers are under constant attack by the various manifestations of global warming. It is important for us: the consumers, the farmers, and the enthusiasts to all be vigilant of and respect the environment of the oyster, and all marine life.
I have created a number of articles on this page that are meant to serve as primers of the various environmental challenges present in the aquaculture industry. My goal is to first draw awareness to the issues plaguing our industry, and also, with assistance of leaders in the scientific and aquaculture communities, offer ways that we can help fight against a depletion of our precious natural resources.
Harmful Algal Blooms
The environment in which an oyster grows greatly influences its appearance, meatiness, and taste. While the environment positively contributes to an oyster’s uniqueness, it oftentimes brings challenges and even threats to the bivalve, its farmer, and its consumers. Throughout history, there have been harmful algal blooms (HABs), which occur when colonies of algae suddenly grow out of control. The rapid accumulation of algal blossom can be significant enough to cause a green, red or brown discoloration of estuarine and freshwater environments. However, not all HABs alter the color of the water, thus making their presence more difficult to detect.
Not all algal blooms are harmful. However, the specific phytoplankton responsible for HABs produces neurotoxins, which are absorbed by shellfish, who are filter feeders. When humans eat the contaminated shellfish, they can suffer from serious, even fatal, illnesses. HABs have also been known to kill fish species, and can also even make the surrounding air difficult to breathe. The presence of HABs necessitates closure of the body of water affected, as well as a ban on fishing and shellfish harvesting. HABs have been reported in every U.S. coastal state, and their occurrences are on the rise. HABs are a national concern because they affect not only the health of the people and marine ecosystems, but also local and regional economies dependent on fishing and aquaculture.
Phytoplankton: The Culprits Behind harmful algal blooms
Phytoplankton are photosynthesizing microscopic organisms that inhabit the upper sunlit layer of almost all bodies of water. Phytoplankton serve as the base of the aquatic food web. Most phytoplankton are too small to be individually seen with the unaided eye. However, when present in high enough numbers, some varieties may be noticeable as colored patches on the water surface due to the presence of chlorophyll.
It is normal for some biotoxin-producing phytoplankton to be present in marine water. They are usually at very low concentrations, and pose no problems. However, certain phytoplankton in increased concentrations can create issues. The potentially-problematic phytoplankton are as follows:
Alexandrium: This phytoplankton produces a toxin that causes Paralytic Shellfish Poisoning.
Pseudo-nitzschia: This phytoplankton produces a toxin that causes Amnesic Shellfish Poisoning.
Dinophysis: This phytoplankton produces a toxin that causes Diarrhetic Shellfish Poisoning.
How Harmful Algal Blooms Occur
The magnitude and severity of HABs, and the subsequent need for shellfish harvesting closures to protect human health, vary considerably from year to year and between decades.
So how do Harmful Algal Blooms occur?
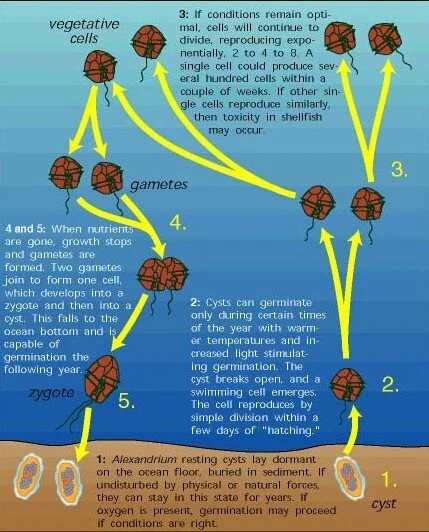
During winter months, some phytoplankton cells turn into cysts which lie dormant in the ocean sediment. When water temperatures warm, the cysts begin to germinate (hatch). After germination, the swimming form of the cell emerges and divides. When environmental conditions are right (oxygen, light, temperature, etc.), cells continue to reproduce and become concentrated, creating blooms. These concentrated blooms release toxins.
The Dangers of Harmful Algal Blooms
The danger of HABs is twofold: (1) they can cause serious illnesses, and even death, in individuals who consume contaminated shellfish; (2) when there is a harmful algal bloom, there is a ban placed on shellfish harvesting in surrounding waters, which affects the aquaculture industry.
Danger to Humans
When algal blooms release toxins, these toxic cells are consumed by oysters that feed by filtering tiny organisms from the water. After the cells are digested, the toxins remain behind, and can accumulate in the tissue of the oyster as more toxic phytoplankton are ingested. This toxin is transferred through the food web when other species, including humans, eat the toxic shellfish. Notably, these toxins are water-soluble and heat- and acid-stable – they are unaltered by ordinary cooking methods.
In Maine, harmful algae blooms typically occur between April and October, when the water temperatures rise. Recently, however, the pseudo-nitzschiai phytoplankton has been blooming in winter months. Thus, unfortunately, these marine toxins can be present in the ecosystem at any time of the year.
Danger to the Aquaculture Economy
In order to keep the public safe, oftentimes, regions must enact precautionary closures of the coastline to prevent shellfish harvesting, and commercial fishing. Historically, HABs generally last only a few weeks and most shellfish (with the exception of some species of clams and scallops which retain the toxin for longer periods) clear themselves rapidly of the toxin once the bloom dissipates.
Recently, however, HABs have been increasing in frequency, and have begun to have more devastating effects on marine life. For instance, there was a significant HAB that occurred in Southwest Florida in 2018 resulting in over 6 tons of dead fish. In July 2019, the Casco Bay region of Maine suffered an HAB that shut down shellfish harvesting for over 10 weeks. Quahog Bay Conservancy is home to Snow Island Oysters. According to Quahog Bay Conservancy’s Development Coordinator, Nicole Twohig, and Consulting Marine Scientist, Darcie Couture, approximately 70,00 oysters could not be sold during that time. According to their calculations, that approximated to $60,000 in lost sales.
When shellfish harvesters are unable to sell their products for weeks at a time, this has an obvious detrimental effect to their business, and, for many, the livelihood of them and their families.
Water Monitoring to Prepare for Harmful Algal Blooms
Scientists at the National Ocean Service have been monitoring and studying this phenomenon for a number of years to determine how to detect and forecast the location of the blooms. The goal is to give communities advance warnings so they can adequately plan for and deal with the adverse environmental and health effects associated with these HABs.
The testing system in place by states affected by HABs is promulgated by the National Shellfish Sanitation Program (NSSP), a federal/state cooperative program recognized by the U.S. Food and Drug Administration (FDA). The purpose of the NSSP is to promote and improve the sanitation of shellfish (oysters, clams, mussels and scallops) moving in interstate commerce through federal/state cooperation and uniformity of State shellfish programs. All shellfish producing States or MOU countries must have a contingency plan that defines administrative procedures, laboratory support, sample collection procedures, and patrol procedures to be implemented on an emergency basis in the event of the occurrence of shellfish toxins.
Water Monitoring to Assess for HABs
The State of Maine, Department of Marine Resources Public Health Bureau tests coastal shellfish areas for biotoxins weekly, beginning in March and going through October, or later when necessary. When shellfish samples are shown to have toxins in concentrations specified by the NSSP, areas are closed to shellfish harvesting.
HAB Forecasts
For the past 12 years, the National Oceanic and Atmospheric Administration (NOAA), in partnership with the National Centers for Coastal Ocean Science (NCCOS) has been producing a yearly HAB forecast for the Gulf of Maine that helps shellfish farmers gauge the severity of the season’s harmful algal blooms before they occur. The forecast is based on data collected about Alexandrium’s dormant stage, when it exists as seed-like cysts in ocean sediments before growing in the spring. Scientists can predict the size of HABs in the spring and summer by counting the number of cysts in bottom sediments the preceding fall.
Throughout the next spring and summer, the NCCOS will provide coastal managers with weekly updates of modeled bloom extent, trajectory, and intensity to complement the seasonal forecast and guide states that will monitor Alexandrium cells and shellfish toxicity along the shore.
To find out whether it is safe to harvest shellfish in a particular area, visit your State’s Department of Marine Resources website. An example of Maine’s DMR Shellfish Closure page, can be found here.
For cited material and further reading, please see:
Harmful Algal Blooms: Tiny Organisms with a Toxic Punch, National Ocean Service.
HAB Monitoring Products: Algal Bloom Monitoring System, developed by the NCCOS.
Maine Biotoxin Closures: Shellfish Biotoxin Inventory: Maine Department of Marine Resources.
Model Ordinance and Standardization Field Guide: National Shellfish Sanitation Program (NSSP).
Longer ‘red tide’ outbreak affecting Maine’s Shellfish Industry, Ryan Breton, News Center Maine, 07/23/2019.
Ocean Acidification
The Earth’s climate has changed throughout history. The “Modern Climate Era” that we experience today began about 7,000 years ago after the end of the last Ice Age. Before that time, there have been seven cycles (!) of glacial advance and retreat that occurred over a 650,000 year period.
Why did these periods of glacial progression happen? Well, up until the Modern Climate Era, these climate changes were attributed to very small changes in the Earth’s orbit. When Earth’s orbit changes, Earth’s distance from the Sun changes. This, in turn, varies the amount of solar energy Earth receives.
So how is the Modern Era different?
The climate change the Earth, and consequently, our oceans, is facing now has nothing to do with a change in the Earth’s orbit. It has to do with us – humans. Further, while the past cycles of climate change produced significant change, they did so over thousands of years. The current warming trend is of particular significance because it is proceeding at a rate that is unprecedented.
The signs of climate change are readily apparent. Turn on any TV or peruse Apple News, and you’ll find reference to rising atmospheric and ocean temperatures, shrinking ice sheets, and glacial retreats.
There is one more effect of climate change that has received increasing attention in the last 10 years – Ocean Acidification. In fact, the term “Ocean Acidification” was not even coined until 2006, which demonstrates just how new the issue is.
What is Ocean Acidification?
Ocean acidification is the ongoing decrease in the pH of the Earth’s oceans caused by the uptake of carbon dioxide (Co2) from the atmosphere. To translate: there is too much carbon dioxide in the water.
Let’s unpack this. For years, the oceans have always absorbed and spewed out carbon dioxide, cycling carbon dioxide back and forth from atmosphere to water. This exchange occurred slowly, over thousands of years. Humans have disturbed this slow exchange by adding some 400 billion tons of carbon to the atmosphere since the middle of the 18th century. Our oceans absorb approximately 30% of the carbon dioxide (Co2) that is released in the atmosphere. So it makes sense that as more Co2 is released into the atmosphere, the levels of Co2 are also rising in our oceans.
More Co2 in the atmosphere means more Co2 being sucked up by the ocean. It has been estimated that approximately 30% of all extra carbon dioxide added to the atmosphere by humans has percolated down into the oceans. In fact, it has been estimated that the amount of carbon dioxide absorbed by the upper layer of the oceans is increasing by about 2 billion tons per year.
How Does an Increase of Co2 in the Ocean Affect Its pH Level?
pH is a scale used to specify how acidic or basic a water-based solution is. Acidic solutions have a lower pH (3.5 – 6.6); basic solutions have a higher pH (7.4-9.0). Think about a lemon – lemon juice has a puckering pH level around 2 (very acidic). Now think about milk – this has a pH level of 6 (neutral). Soap, and many household cleaning products have a pH level of 10 (very basic).
The pH of seawater has hovered around 8.2 on the pH scale for hundreds of years– thus, seawater has always been slightly basic.
Now don’t get it wrong – the pH level of seawater has changed over the past hundreds of thousands of years. During cold stages in the planet’s past, the pH level crept up (became more basic) by something like 0.2 pH units, and it crept down (became more acidic) by about the same amount when the planet warmed up. But it took tens of thousands of years for those changes to happen—plenty of time for creatures living in the seas to adjust to the change.
In the past 250 years, however, the oceans’ pH levels have changed more than they have in tens of thousands of years. Since the Industrial Revolution, the ocean’s pH level has fallen by 0.1 pH units, and thus, has become more acidic. This means that the oceans are now 30% more acidic today than they were a short 250 years ago.
How Did This pH Shift Happen?
Under normal conditions, there is an abundance of free-floating “pure” carbonate ions in ocean water. However, when Co2 is absorbed by seawater, a series of chemical reactions occur, resulting in the increased concentration of hydrogen ions. These added hydrogen ions bind to the free-floating “pure” carbonate ions, forming a molecule called bicarbonate (HCO3). When this happens, there is not enough “pure” carbonate ions left in the water.
Guess what shellfish shells are made out of? Carbonate.
How Does Ocean Acidification Affect Oysters and Other Shellfish?
An analogy for you: ocean acidification is a lot like osteoporosis. Oysters, and other shellfish, rely on carbonate ions to help build and strengthen their shells. In a normal ocean, one carbonate ion is taken from the water and combined with calcium atom to form the shells of many animals. In an acidic ocean, abundant hydrogen ions bind with carbonate and prevent shell formation. The increasingly lower pH of ocean water means there is decreasing carbon. Thus, shellfish do not have the essential components to strengthen – and, in the case of larvae, even build - their shells.
Larvae Formation: Oyster larvae don’t have shells when they are spawned. It takes approximately 12-24 hours after being spawned for oysters to build a shell. Larvae need to gather enough calcium and carbonate from the water to create a shell. They are just babies, and have very little time and energy to do this. If they cannot form a shell due to the lack of carbonate ions in the water, they die.
Stunted Growth: While acidification impacts oysters at the larval stage, acidification also affects adult oysters. Adult oysters continue to add more shell as they grow, but if carbonate is unavailable, the oysters must expend additional energy to add shell. If their energy is going towards building shell that is continually being eroded, it cannot go towards growing larger. Therefore, oysters being stressed by ocean acidification will not be as large as those growing in a healthier environment.
Weakening Shells: An excess of hydrogen ions in the water can actually attack the calcium carbonate in existing oyster shells, causing them to break down. In fact, scientists are actually predicting that if ocean acidification continues at this rate, by 2100, calcium carbonate shells may begin dissolving altogether.
Predators: An oyster’s shell protects it from ocean predators, such as fish or sea sponges. If an oyster’s shell is weakened, it can be broken by predators.
Destruction of Oyster Habitats and Oyster Reefs: In the wild, baby oysters need to settle on a hard substrate, such as a rock or an old oyster reef, to grow. The older shells which formed the foundation of the reef are now being dissolved by ocean acidification.
Not only do shells protect oysters from predators in their environment, but the shells also allow the oysters to develop internally and grow their meatiness and flavor profile.
Where Do We Go From Here?
This article is meant to serve as a basic primer on a very complex issue in aquaculture. I will be drafting a series of follow-up articles, based on conversations with environmental researchers and advocates for climate change to provide further information on what is being done to combat this very serious threat. For additional reading and education, I recommend the following: